Recently I had a discussion on LinkedIn about the pump killer #2: ‘wrong material’. There I chipped in with this disaster picture1. It was an application where we provided a suboptimal material for the acid environment. The consequences were disastrous, as seen above. Luckily, we were able to identify the problem and propose a different material. Now, I want to share our experience here, also.
What was the case? A client requested a DOP for handling tailings in their facility. Tailings is fine stuff. Leftover from mining or waste water processing. We are always careful on the grain size, as these fines may interfere with the operation of the mechanical seal. With appropriate measures, they can handle them. As the grains tend to be fresh, they can be razor sharp. The erosion on the wear parts is higher than normal fine silt. Oh, and most tailings come with acid in their water.
So, for this request we proposed a material that was usually good in wear resistance and had a moderate resistance against corrosion. Casting materials can be classified for their corrosion resistance with the Pitting Resistance Equivalent Number2. This PREN can be calculated with:
PREN = Cr + 3.3Mo + 16N
Two observations to this formula. One, this is only valid for normal Chromium content materials intended as Stainless Steel. Two, it does not mention the aggression of the corrosion. The acidity is usually provided in the request for quotation. But, a catalyst for the oxidising reaction is the conductivity of the fluid. Chloric acid and sulphuric acid may have the same pH, but due to their different ion and electron content, their conductivity differ. We did not check this in the above example, with the consequences in the picture.
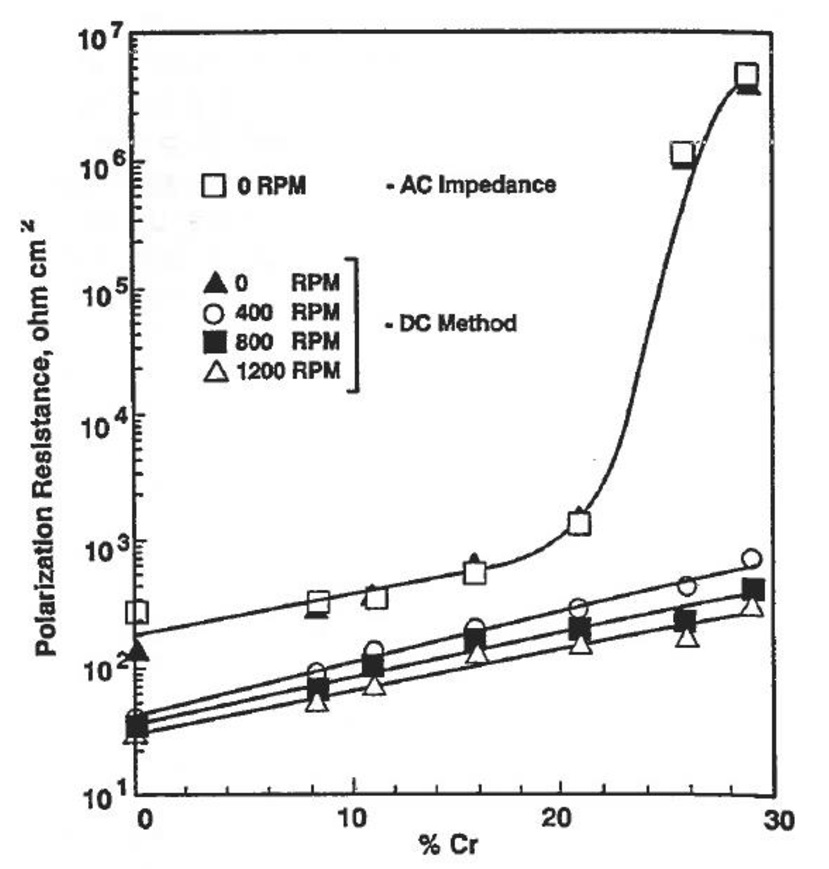
Is increasing the chromium content in the wear alloy a solution to this corrosion problem? Mwah, moderately. Alloys like stainless steel profit from the above approach. But, wear materials use their Chromium for generating carbides. Those are the particles we require for the wear resistance. The Chromium provided is than not available for corrosion resistance. e.g. White cast iron with 3%C and 21%Cr will only have about 6% of Chromium to be used in the PREN. For white cast irons, it is better to use the following graph3 to find their corrosion resistance.
If corrosion is such an issue, why don’t we use Stainless Steel? Well, there you bite yourself in the tail. Stainless Steel in itself is relatively soft. It would have the same wear index of normal construction steel. By definition a Wear Rate Index of 1. For the sharp tailing material, that would be disastrous in itself. But, let’s play along. The stainless steel derives it’s corrosion resistance from the Chromium as explained before. Chromium’s trick is to generate a clear closed patina layer of Chromium Oxide protecting the underlaying material. In dredging conditions, the particles damage the protecting patina forever exposing fresh base material for more erosion and corrosion. In the end, the wear is accelerated and part life decreases dramatically.

Back to the pictured example, we expected some corrosion, but did not expect the higher conductivity. So, after three weeks, the client noticed a sudden los of performance. The leading edge of blades and the hub shroud were completely eaten away. As long as the trailing edge was there, it generated head. A single stone hit severed the front of the impeller from the hub and we received the above disaster picture. After damage evaluation, we sent a CW250 impeller and that one lasted.

References
- Pump killers: How to fight the 13 most common centrifugal pump failures? Number 2., Jos Overschie
- Pitting resistance equivalent number, Wikipedia
- Corrosion Properties of Cast Iron Ball Materials in Wet Grinding, Corrosion